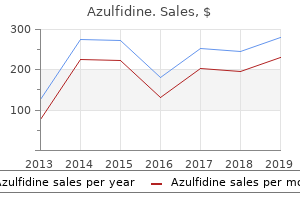
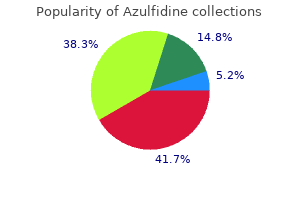
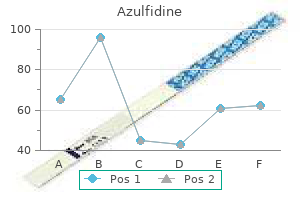
"Generic azulfidine 500mg without a prescription, cape fear pain treatment center dr gootman".
By: J. Candela, M.A., M.D.
Medical Instructor, University of Cincinnati College of Medicine
Srisurapanont M pain treatment kidney stone order azulfidine with mastercard, Jarusuraisin N phantom pain treatment buy azulfidine discount, Kittirattanapaiboon P: Treatment for amphetamine dependence and abuse pain and injury treatment center purchase 500mg azulfidine free shipping. Yui K unifour pain treatment center hickory nc purchase 500mg azulfidine amex, Goto K, Ikemoto S: the role of noradrenergic and dopaminergic hyperactivity in the development of spontaneous recurrence of methamphetamine psychosis and susceptibility to episode recurrence. Moscovitz H, Brookoff D, Nelson L: A randomized trial of bromocriptine for cocaine users presenting to the emergency department. Wolfsohn R, Angrist B: A pilot trial of levodopa/carbidopa in early cocaine abstinence. Kosten T, Oliveto A, Feingold A, Poling J, Sevarino K, McCance-Katz E, Stine S, Gonzalez G, Gonsai K: Desipramine and contingency management for cocaine and opiate dependence in buprenorphine maintained patients. Ling W, Shoptaw S, Majewska D: Baclofen as a cocaine anti-craving medication: a preliminary clinical study. Konefal J, Duncan R, Clemence C: the impact of the addition of an acupuncture treatment program to an existing metro-Dade County outpatient substance abuse treatment facility. Shwartz M, Saitz R, Mulvey K, Brannigan P: the value of acupuncture detoxification programs in a substance abuse treatment system. Rosenblum A, Magura S, Palij M, Foote J, Handelsman L, Stimmel B: Enhanced treatment outcomes for cocaine-using methadone patients. Schiffer F: Psychotherapy of nine successfully treated cocaine abusers: techniques and dynamics. Kranzler H, Rounsaville B: Dual Diagnosis: Substance Abuse and Comorbid Medical and Psychiatric Disorders. Shesser R, Davis C, Edelstein S: Pneumomediastinum and pneumothorax after inhaling alkaloidal cocaine. Delaney-Black V, Roumell N, Shankaran S, Bedard M: Maternal cocaine use and infant outcomes (abstract). Office of National Drug Control Policy: Drug Policy Information Clearinghouse Fact Sheet: Heroin. Farre M, Mas A, Torrens M, Moreno V, Cami J: Retention rate and illicit opioid use during methadone maintenance interventions: a meta-analysis. Uchtenhagen A: Swiss Methadone Report: Narcotic Substitution in the Treatment of Heroin Addicts in Switzerland. Farrell M: A Review of the Legislation, Regulation and Delivery of Methadone in 12 Member States of the European Union: Final Report. Luxembourg, Office for Official Publications of the European Communities, 1996 [G] 1344. Maremmani I, Zolesi O, Aglietti M, Marini G, Tagliamonte A, Shinderman M, Maxwell S: Methadone dose and retention during treatment of heroin addicts with axis I psychiatric comorbidity. Maremmani I, Canoniero S, Pacini M: Methadone dose and retention in treatment of heroin addicts with bipolar I disorder comorbidity: preliminary results. Langrod J, Lowinson J, Ruiz P: Methadone treatment and physical complaints: a clinical analysis. Lexington, Ky, National Academy of Sciences, National Research Council, 1967 [G] 1357. Darke S, Sims J, McDonald S, Wickes W: Cognitive impairment among methadone maintenance patients. Cami J, de Torres S, San L, Sole A, Guerra D, Ugena B: Efficacy of clonidine and of methadone in the rapid detoxification of patients dependent on heroin. Spencer L, Gregory M: Clonidine transdermal patches for use in outpatient opiate withdrawal.
Prescribing health care professionals should be familiar with these side effects and take them into consideration before prescribing pain management with shingles cost of azulfidine. Thus pain treatment a historical overview purchase genuine azulfidine on-line, once disulfiram is taken by mouth pain treatment ibs generic azulfidine 500mg online, any alcohol consumed results in rapid buildup of acetaldehyde and a negative reaction or sickness results treatment for long term shingles pain generic 500mg azulfidine with mastercard. The intensity of this reaction is dependent on the dose of disulfiram and the amount of alcohol consumed. Disulfiram is most effective when its use is supervised or observed, which has been found to increase compliance. Thus, an individual who wants to reduce, but not stop, drinking is not a candidate for disulfiram. Because it blocks some opioid receptors, naltrexone counteracts some of the pleasurable aspects of drinking. Many studies have examined the effectiveness of naltrexone in treating alcohol use disorders. Adherence to taking the medication increases under conditions where it is administered and observed by a trusted family member or when the extended-release injectable, which requires only a single monthly injection, is used. These therapies also teach and motivate patients in how to change their behaviors as a way to control their substance use disorders. Despite this, many counselors and therapists working in substance use disorder treatment programs have not been trained to provide evidence-based behavioral therapies, and general group counseling remains the major form of behavioral intervention available in most treatment programs. These therapies have been studied extensively, have a well-supported evidence base indicating their effectiveness, and have been broadly applied across many types of substance use disorders and across ages, sexes, and racial and ethnic groups. Individual counseling is delivered in structured sessions to help patients reduce substance use and improve function by developing effective coping strategies and life skills. Most studies support the use of individual counseling as an effective intervention for individuals with substance use disorders. These sessions typically explore the positive and negative consequences of substance use, and they use self-monitoring as a mechanism to recognize cravings and other situations that may lead the individual to relapse. Contingency management, which involves giving tangible rewards to individuals to support positive behavior change,85 has been found to be effective in treating substance use disorders. A group providing mutual support and fellowship for people recovering from addictive behaviors. All three treatments reduced the quantity and frequency of alcohol use immediately after treatment. Studies of various family therapies have demonstrated positive findings for both adults and adolescents. In a recent review of controlled studies with alcohol-dependent patients, marital and family therapy, and particularly behavioral couples therapy, was significantly more effective than individual treatments at inducing and sustaining abstinence; improving relationship functioning and reducing intimate partner violence; and reducing emotional problems of children. Research has shown that incorporating tobacco cessation programs into substance use disorder treatment does not jeopardize treatment outcomes212 and is associated with a 25 percent increase in the likelihood of maintaining long-term abstinence from alcohol and drug misuse. These supportive services are typically delivered by trained case managers, recovery coaches, and/or peers. Specific supports include help with navigating systems of care, removing barriers to recovery, staying engaged in the recovery process, and providing a social context for individuals to engage in community living without substance use. Further, active recovery and social supports, both during and following treatment, are important in maintaining recovery. The use of telehealth to deliver health care, provide health information or education, and monitor the effects of care, has also rapidly increased. It offers alternative, cost-effective care options for individuals living in rural or remote areas or when physically travelling to a health care facility poses significant challenges. Telemedicine refers specifically to remote clinical services, whereas telehealth can include remote nonclinical services such as provider training, administrative meetings, and continuing medical education, and patient-focused technologies, in addition to clinical 1 services. They can increase access to care in underserved areas and settings; free up time so that service providers can care for more clients; provide alternative care options for individuals hesitant to seek in-person treatment; increase the chances that interventions will be delivered as they were designed and intended to be delivered; and decrease costs. After 60 days, members of intervention group with alcohol dependence reported significantly fewer drinks per drinking day as compared to control group (3. Lower rates of drinks per drinking day among intervention group maintained at 12-month follow-up.
Opioid addiction often pain joint treatment order azulfidine australia, but not always unifour pain treatment center statesville order azulfidine with a mastercard, is accompanied by tolerance wnc pain treatment center arden nc order on line azulfidine, physical dependence heel pain treatment stretches buy azulfidine in india, and opioid withdrawal symptoms. Opioids that bind to opioid receptors but block them, rather than activating them, are termed opioid antagonists. Opioid partial agonists are drugs that activate receptors, but not to the same degree as full agonists. Increasing the dose of a partial agonist does not produce as great an effect as does increasing the dose of a full agonist. The agonist effects of a partial agonist reach a ceiling at moderate doses and do not increase from that point, even with increases in dosage. It is the partial agonist properties of buprenorphine that make it a safe and an effective option for the treatment of opioid addiction. Buprenorphine has sufficient agonist properties such that when it is administered to individuals who are not opioid dependent but Executive Summary xvii who are familiar with the effects of opioids, they experience subjectively positive opioid effects. These subjective effects aid in maintaining compliance with buprenorphine dosing in patients who are opioid dependent. Buprenorphine occupies opioid receptors with great affinity and thus blocks opioid full agonists from exerting their effects. This enables daily or less frequent dosing of buprenorphine, as infrequently as three times per week in some studies. Buprenorphine is abusable, consistent with its agonist action at opioid receptors. Its abuse potential, however, is lower in comparison with that of opioid full agonists. A formulation containing buprenorphine in combination with naloxone has been developed to decrease the potential for abuse via the injection route. Physicians who prescribe or dispense buprenorphine or buprenorphine/naloxone should monitor for diversion of the medications. Screening the Clinical Guidelines for the Use of Buprenorphine in the Treatment of Opioid Addiction Consensus Panel recommends that physicians periodically and regularly screen all patients for substance use and substancerelated problems, not just those patients who fit the stereotypical picture of addiction. The full text of selected screening instruments is provided in Appendix B, Assessment and Screening Instruments. Complete assessment may require several office visits, but initial treatment should not be delayed during this period. The Guidelines document provides recommendations on effective interviewing techniques and on the components of the complete history, physical examination, and recommended initial laboratory evaluation of patients with opioid addiction. The consensus panel recommends that initial and ongoing drug screening should be used to detect or confirm the recent use of drugs. Urine screening is the most commonly used and generally most cost-effective testing method. Chapter 3, Patient Assessment this chapter provides an approach to the screening, assessment, and diagnosis of opioid addiction problems, and for determining when buprenorphine is an appropriate option for treatment. The necessary first steps in the medical management of opioid addiction are (1) the use of validated screening tools to identify patients who may have an opioid use problem and (2) further assessment to clearly delineate the scope of an opioid addiction problem when one is identified. When treatment is indicated, consideration must be given to the appropriate treatment approach, xviii Executive Summary Diagnosis of Opioid-Related Disorders After a thorough assessment of a patient has been conducted, a formal diagnosis can be made. This diagnosis is based not merely on physical dependence on opioids but rather on opioid addiction with compulsive use despite harm. The evaluation includes determining if appropriate patient motivation exists and ruling out contraindicating medical and psychiatric comorbidities. A variety of clinical scenarios are addressed, including whether patients are addicted to long- versus shortacting opioids, and whether the approach selected is maintenance treatment or medically supervised withdrawal (which must be followed by long-term drug-free or naltrexone treatment to be useful to the patient). Maintenance Treatment Maintenance treatment with buprenorphine for opioid addiction consists of three phases: (1) induction, (2) stabilization, and (3) maintenance. Induction is the first stage of buprenorphine treatment and involves helping patients begin the process of switching from the opioid of abuse to buprenorphine. The goal of the induction phase is to find the minimum dose of buprenorphine at which the patient discontinues or markedly diminishes use of other opioids and experiences no Executive Summary xix withdrawal symptoms, minimal or no side effects, and no craving for the drug of abuse.
It also appears to be effective as a withdrawal (detoxification) agent (1674 midsouth pain treatment center reviews generic azulfidine 500mg with visa, 1706) allied pain treatment center youngstown ohio generic azulfidine 500 mg on line. Because of its poor oral bioavailability pain and headache treatment center in manhasset ny best azulfidine 500mg, most clinical trials of buprenorphine for the treatment of opioid dependence have used a sublingual unifour pain treatment center lenoir nc buy azulfidine 500 mg fast delivery, often alcohol-based, solution. A liquid sublingual form of buprenorphine, however, was felt to be an impractical long-term treatment option, and as the evidence for its efficacy and safety in the treatment of opioid dependence grew, a watersoluble sublingual tablet form of buprenorphine was developed. For this reason, a formulation that combines buprenorphine with naloxone was developed for the U. Naloxone has poor sublingual bioavailability (1361), so use of this combination tablet sublingually would produce a predominant buprenorphine effect. Although the combination tablet is the more commonly used form of this medication, many clinical trials have used a monotherapy tablet, and the monotherapy tablet is the primary form of sublingual buprenorphine used outside the United States. Under chronic dosing conditions, there may be slightly better bioavailability for the combination versus the monotherapy tablets (1710). However, for a given dose of buprenorphine, there is considerable between-subject variability in buprenorphine blood levels. Use of buprenorphine on a daily basis as a maintenance agent Numerous randomized, double-blind clinical trials have studied the efficacy and safety of sublingual buprenorphine for the outpatient treatment of opioid dependence. This section reviews the three representative studies that compared buprenorphine with placebo and then provides a more limited review of the many studies that compared buprenorphine with methadone. The first study was a 16-week multisite double-blind, randomized outpatient clinical trial using four different doses of sublingual buprenorphine solution: 1, 4, 8, and 16 mg/day (1725). The primary goal was to compare the 8- and 1-mg doses, with the 1-mg dose serving as the placebo condition. The study enrolled 736 opioid-dependent patients (about 33% female); primary outcome measures were treatment retention, opioid urinalysis results, craving, and global ratings by staff and patients. Results from the study showed significantly better outcomes for the 8- versus the 1-mg groups on all the primary outcome measures. In general, there was no clear pattern of increased side effects or adverse events for the 8-mg group, with the exception of ratings of constipation (but multiple comparisons were made). Although not the primary purpose of the study, the outcomes also showed dose-related effects for buprenorphine, where 4 mg of buprenorphine was better than 1 mg, 8 mg was better than 4 mg, and 16 mg was better than 8 mg, although these differences were not always robust in their magnitude. The second study was a double-blind, placebo-controlled study that used a somewhat novel clinical trial design that lasted only 2 weeks (1726). In this study, the 150 male and female participants were fast-tracked into treatment and randomly assigned to 0 (N=60), 2 (N=60), or 8 (N=30) mg/day of sublingual buprenorphine solution. Subjects were informed that they could receive placebo or one of two buprenorphine doses and that after 6 days, they could request to have a blind change to one of the other two conditions. Primary outcomes were the percentage of patients in each condition who remained on their original dose and the percentage of patients in each condition who requested a dose change. Other study outcomes such as opioid urine test results and self-reports of drug use were also provided. This study found that, regardless of dose, a significantly higher percentage of patients in the two active conditions remained on their doses compared with the placebo group. Similarly, a significantly higher percentage of patients in the placebo condition requested a dose change compared with the other two groups, but, once again, there was no significant difference between the two active buprenorphine groups. In an interesting finding, male subjects in the two active buprenorphine groups had a significantly lower rate of opioid-positive urine samples, but there was no difference across the three conditions for female subjects. This study provides an alternative demonstration of the efficacy of buprenorphine compared with placebo. The third study was an office-based protocol that compared sublingual placebo tablets to active buprenorphine/naloxone (16 mg/4 mg) and buprenorphine alone (16 mg) tablets (1727). This multicenter study enrolled 326 opioid-dependent individuals, and study participation lasted 4 weeks for each volunteer.
Discount azulfidine 500 mg with amex. Stomach Pain Relief- Alternative Healing-Pure Tone Brainwave Entrainment.



